Role of Alloimmunity in Pre-Eclampsia
Introduction
Pre-eclampsia is a pregnancy-specific heterogenous vascular disease of the human pregnancy. It usually presents in a woman who is previously normotensive, during the second half of pregnancy and is characterised by hypertension, proteinuria and edema or pre-eclampsia associated signs without proteinuria. Pre-eclampsia is observed in 3 – 8% of pregnancies. It is one of the most significant causes of maternal and fetal morbidity and mortality worldwide. There is increasing evidence now that implicates important role of alloimmune problem in the etiopathogenesis of pre-eclampsia.
Immuno-physiology of Pregnancy
Immune system plays an important role in the maintenance of pregnancy. Maternal immune response during pregnancy is essential to maintain pregnancy. The fetus possesses half maternal genes and half paternal genes (or from donor male), making it a ‘semi-allograft’ in the immunologically active mother. In case of donor egg or donor embryo, the pregnancy is a complete allograft. However, the fetus grows unabated in the mother’s body due to development of immune tolerance by the mother’s immune system. The more genetically distinct the fetus is, the greater is the immunological tolerance during pregnancy. This is known as the ‘paradox’ of pregnancy.
Immunopathology of Pre-eclampsia
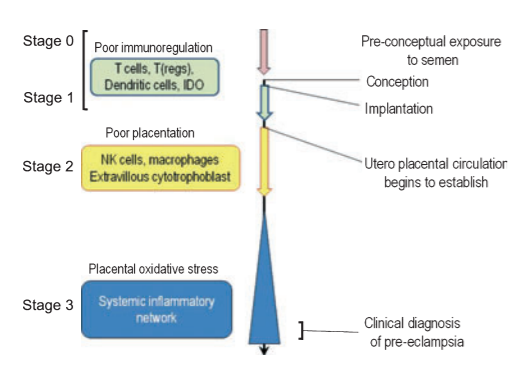
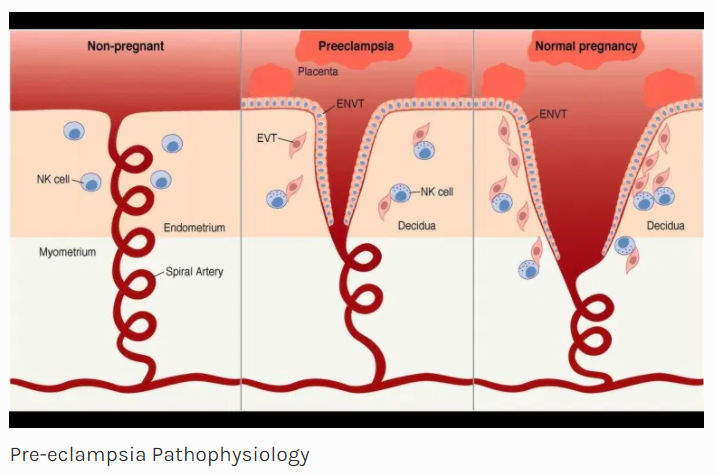
Pre-eclampsia is a pregnancy specific disorder characterised by hypertension and proteinuria and may progress to a multiorgan disease. The exact etiology of pre-eclampsia is still unknown. However, deficient early placentation is particularly associated with early onset pre-eclampsia and this abnormal placentation is thought to be immunologically mediated. It is likely that pre-eclampsia results from abnormal trophoblast growth and differentiation at any time after the earliest stages of implantation. The primary dysfunction may be immunological leading to three stages of pre-eclampsia.
Immunologic Stages of Pre-eclampsia
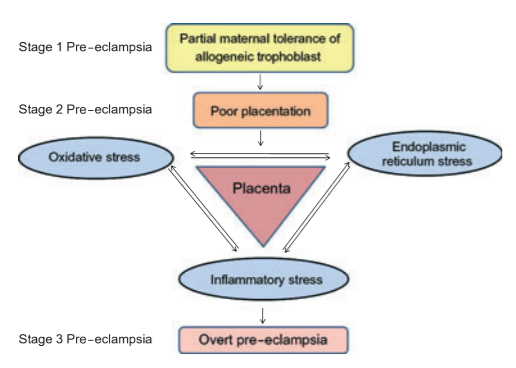
According to the immunological changes occurring at the fetomaternal interface, Pre-eclampsia is divided into 3 stages:
(Some consider presence of Stage 0 in the development of pre-eclampsia. This stage is before conception. The absence of seminal plasma predisposes to non-development of maternal tolerance to paternal antigens.)
Stage I:
This is seen in very early pregnancy at 3-8 weeks of gestation. This stage involves implantation and very early placentation (Histiotrophic placenta). During normal pregnancy, there are Immunoregulatory mechanisms at the fetomaternal interface which help in continuation of pregnancy. This involves interaction between decidual immune cells (T – Cells, Macrophages, and dendritic cells) and class I HLA (mainly C) from the trophoblast. This happens due to allo-recognition. If there is total failure of immunoregulatory mechanisms, it leads to pregnancy loss. However, a partial failure leads to continuation of pregnancy with defective placentation process. This creates a continuum between abortion due to allo-immune cause and pre-eclampsia.
In this process, tryptophan plays an important role. Indolamine 2, 3- dioxygenase (IDO) enzyme, present in decidual immune cells leads to depletion of tryptophan by its catabolism. Depletion of tryptophan promotes differentiation of Th17 cells to Treg cells, which produce immunotolerance and continuation of pregnancy. Failure of activation of IDO results in pregnancy loss but dysregulation of IDO results in poor placentation and paves way to pre-eclampsia. These are no clinical symptoms or signs in this stage.
Stage II:
This occurs in late 1st trimester and early second trimester (8-18weeks). This stage mainly involves abnormalities in placentation and there is defective formative of cytotrophoblast (chorion frondosum and chorion laeve) and syncytiotrophoblast. There is a second wave of placentation at this time and the dysregulation of immune mechanism between NK cells from decidua and HLA-C from trophoblast continues. These are no clinical signs and symptoms during this stage.
Stage III:
This is seen in late second trimester and in third trimester. This occurs post placentation and it involves interaction between cytotrophoblast (chorior laeve) & villous syncytiotrophoblast on one side and decidua & maternal blood on the other side.
The poor placentation that has resulted due to events in stages I & II leads to spasm of small muscular spiral arteries leading to high pressure pulsatile flow to intervillous space. Rapid changes in oxygenation and hydrostatic stress lead to endothelial damage. The resulting oxidative and endoplasmic reticulum stress leads to inflammatory changes. In this stage, the disease process goes beyond rejection of “allogenic placenta” and is characterized by a global maternal inflammatory response to a damaged placenta. The inflammatory Th1 environment further suppresses generation of Treg cells leading to loss of immune tolerance.
Role of trophoblasts derived factors in pre-eclampsia
The maternal systemic inflammation, which is the hallmark of pre-eclampsia, is contributed by many trophoblast-derived factors like activin-A, corticotrophin releasing factor, leptin & free heme. However, the most important factor is the micro-vesicles and nanovesicles released by the activated placental cells. There is significantly higher amount of micro- and Nanovesicles seen in pre-eclampsia. They are pro-inflammatory and have an anti-endothelial effect.
The micro-vesicular shedding from the syncytiotrophoblast increases in two situations. The first is when the placental size is large, as seen in twin gestation or large placenta in late third trimester. In this situation the onset of pre-eclampsia is in the third trimester. The second situation is when placenta is damaged due to the inflammatory process caused by oxidative stress and endoplasmic reticulum stress. This leads to early onset and more severe form of pre-eclampsia. In this case, the placenta may be abnormally small and associated with severe inflammatory process.
Immunology of Pre-eclampsia and ART
In natural conception, seminal fluid plays an important role in inducing immune tolerance even before conception. Exposure to seminal fluid induces the accumulation Treg cells in the uterus prior to embryo implantation. Treg cells are essential in implantation.
Use of donor spermatozoa increases the risk of pre-eclampsia (18.2%). Risk of pre-eclampsia in IVF and ICSI with ejaculated sperms is the same (4%), but in ICSI with surgically obtained sperms, it is significantly higher (11%). In case of ovum donation (OD), where there is previous exposure to the partner’s semen and the fetus is a semi-allograft, the risk of pre-eclampsia is 16%. However, when embryo donation (ED) is done, there is no previous exposure to the semen and the fetus is complete allograft, the risk of pre eclampsia is very high (33%).
Conclusion
Pre-eclampsia is a dreaded obstetric condition characterised by complex immune mechanisms. It develops in stages. Before conceptions (stage 0), absence of seminal plasma predisposes to non-development of maternal tolerance to paternal antigens. In stage I and II, there is partial failure of immunoregulation leading to poor placentation and abnormalities of utero-placental perfusion. The third stage, which is clinical, is characterised by systemic inflammatory response involving multiple systems. Use of donor gametes increase the chances of development of pre-eclampsia maximum being in ED pregnancies as the fetus is completely allogenic and no seminal exposure.